Lifebit
Case Studies
Discover how leading organizations use Lifebit in our health data case studies — overcoming data challenges and driving research impact at scale.
Governments
Trusted by governments. Proven at scale.
The platform the public sector relies on.

Lifebit turns Genomic Data into Personalised Cancer Treatment
“Lifebit Platform has been a game-changer, driving crucial progress in the breast cancer Research Network.”
Dr Daniella Black
CHALLENGES
Massive data volumes generated per genome
Complex, multi-stage clinico genomic analysis pipelines
High I/O demands and extremely long analyses runtimes
Escalating compute costs
Slow iteration cycles delaying scientific discovery
Accelerated translation from research insight to clinical application
OUTCOMES
Identification of actionable genomic biomarkers in breast cancer
Improved patient stratification for targeted therapies
Faster discovery cycles for genomic research teams
Reduced computational cost for large-scale studies
Accelerated translation from research insight to clinical application
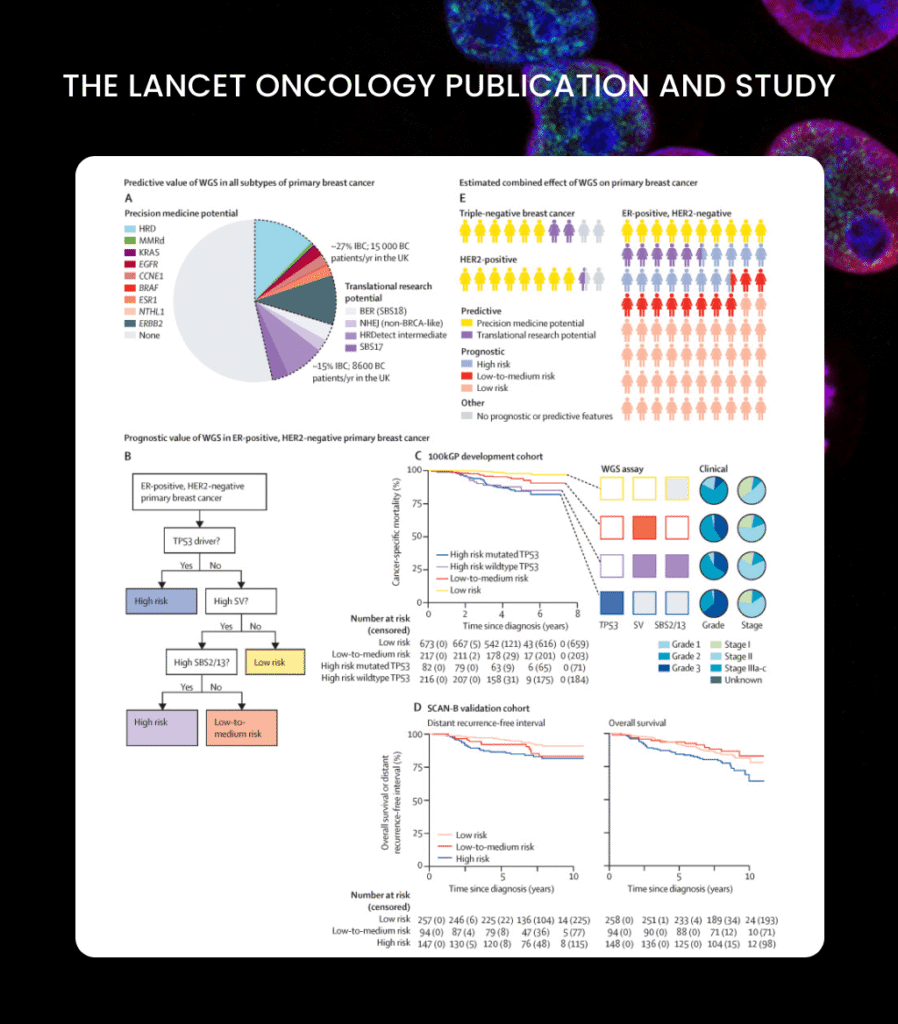
27%
Breast cancer patients
with actionable genomic findings
70%
faster Runtime
for large-scale genomic analysis workflows
2,500+
Patient genomes
analysed across England
50%
lower Compute cost
for population-scale biomarker discovery
15,000+
Women annually
who could benefit from personalised treatment

Lifebit turns Genomic Data into Personalised Cancer Treatment
“With Lifebit’s data transformation and mapping tools, we can process and harmonize distributed data through a single centralized, auditable, and reproducible workflow — something that would normally take years to achieve.”
Consensus feedback from NLM Federated Data Workshop participants
CHALLENGES
Siloed, fragmented data assets both within and between labs and ICs.
Requiring NIH authentication to any cloud-based system
No ability to analyze large distributed cohorts
Limited standardization of data and data elements within or between disease areas
Lack of privacy-preserving methods to identify patients across studies
OUTCOMES
First federated platform ever deployed in NIH
Research authentication via NIH Identity Access Management (IAM)
Collaboration without moving sensitive data—reducing delays and costs.
Reduced privacy and governance risk
Faster scientific discovery through increased intra- and inter-IC data access.

1hr
Data access request
down from 2-weeks
8
ICs onboraded
across NIH
19
pan-IC datasets
mapped to the OMOP CDM
100%
Data standardisation
achieved

Lifebit turns Genomic Data into Personalised Cancer Treatment
“I am incredibly excited that Lifebit joined us to launch the next phase of our research capabilities.”
Chris Wigley
Former-CEO of Genomics England
CHALLENGES
strict privacy and governance requirements
fragmented data environments
high cost and risk of transferring datasets
limited ability to analyse large distributed cohorts ipsum
OUTCOMES
30–90% lower cloud costs through large-scale analysis optimisation
Improved collaboration
Faster scientific discovery through faster access to data
Access to low/no-code user functionality
Easy to bring own tools and external data

125,500
cancer & rare disease
genomes with full clinical data
50
petabytes
of data
1200+
users
30-90%
lower
cloud costs

Lifebit turns Genomic Data into Personalised Cancer Treatment
“It is so inspiring and rewarding to see the next generation of researchers engaging with tools and resources like our dataset and Lifebit’s Trusted Research Environment.”
Vicki Kirsh
Scientific Coordinator
CHALLENGES
Diverse regulatory requirements across eaach of Canada’s provinces
High risk of transferring data sets directly to researchers
Sending new copies of data to researchers creates duplicates and multiple sources of truth
Lack of a standardized analytical environment makes it impossible to teach a core set of analytical skills at scale
No user access controls so one can’t confirm that only approved researchers are accessing the data
OUTCOMES
Centralized data management improves time-to-delivery for research data requests
Differential data access controls ensure the right data is shared without needing to create copies
Platform modularity facilitates alignment to course curricula and workshop learning outcomes
The flexibility of analytical tooling in the platform means workflows can be executed in minutes instead of days or weeks.

100%
Adherence
to pan-Canadian regulatory requirements
140+
Researchers and students
trained in next-gen data analytics in 3 months
50%
Decrease
in time-to-data access and analyses for researchers

Lifebit turns Genomic Data into Personalised Cancer Treatment
“Through our collaboration with Lifebit, we are advancing the use of research and clinical insights using clinico-genomic data for precision medicine. By doing so, we hope to accelerate the translation into clinical workflows, in a manner that will make a significant impact to improving healthcare for Singaporeans.”
Andy Ta
Chief Data Officer and Director, Data Analytics & AI of Synapxe
CHALLENGES
20PB of genomic and linked health data, making data transfer and duplication impractical
High PDPA and MOH security requirements to access sensitive genomic and clinical datasets
Need to scalable support 1000+ researchers and multiple external partners (including pharma)
High I/O demands and extremely long analyses runtimes
High complexity of securely linking multi-modal datasets (genomics, EHR, registries, imaging, lifestyle
OUTCOMES
Federated, privacy-preserving analysis without moving sensitive data
Secure data linkage across genomic, clinical, registry, and longitudinal datasets
Accelerated precision medicine for Asian populations, with faster translation to clinical and population health impact
Improved biomarker discovery and patient stratification across population-scale cohorts
Enablement of public-private partnerships between healthcare, academia, and pharma

100,000
Singaporean participants
in SG100K multi-ethnic cohort (20% Malay, 20% Indian, 60% Chinese)
20
Petabytes
of data
37
Research
projects launched
7
Global
pharmaceutical companies engaged
100%
Adherence
to Singapore Gov. security & regulatory requirements
Commercial
One platform. Every partner. Zero limits.
Built for pharma, providers and nonprofits.

Lifebit turns Genomic Data into Personalised Cancer Treatment
“The advanced capabilities of Lifebit’s Trusted Research Environment, coupled with our focus to reimagine the infrastructure of cancer care, are enabling us to unlock the potential of cancer data.”
Dr. Arun Sujenthiran
UK Clinical Lead and Senior Medical Director
CHALLENGES
Secure, governed analysis across distributed oncology datasets, ensuring compliance with regional data regulations without requiring data centralisation
Scalable data processing and ingestion, reducing the operational burden of reprocessing and validating data through automated pipelines and efficient data refreshes
Federated orchestration across jurisdictions, enabling coordinated updates and analyses across region-specific datasets without moving data
OUTCOMES
Federated data discovery, querrying and analyse across multiple countries
Secure TRE enabling analysis of sensitive patient-level data without moving data across borders, ensuring strict privacy controls and compliance with national regulations.
Improved collaboration
Frequent data ingestion enables faster insights than traditional registries.

5M+
Anonymised
patient records available for researchwith actionable genomic findings
30+
Partners
acros US, UK, Germany and Japan
100%
Federation
across US, UK, Germany and Japan
90
cycle
for data refreshes
Lifebit turns Genomic Data into Personalised Cancer Treatment
“The Lifebit Platform is playing an important part of Boehringer Ingelheim’s broader strategy to capture translational disease insights from large external healthcare biobanks and ultimately to accelerate the development of innovative medicines.”
Dr. Jan Nygaard Jensen
SVP & Global Head of Computational Biology & Digital Sciences
CHALLENGES
Providing secure access to UK Biobank data that would be in-line with the UKB governance policies.
Utilizing the Five Safes Principles to ensure data security and privacy while allowing user acces and analysis.
UK Biobank and other biobank data standardized to OMOP so that they can be querried and analysed jointly.
Organise complex analytical workflows into logical units of work and publish the final results for wider use.
Federated betweein Genomics England, UK Biobank and others.
OUTCOMES
+90% faster target identification and validation analyses
Federated and joint analyses between UK Biobank, Genomics England and other biobank data.
Secure access to approved internal clinical data, with data never leaving the Trusted Research Environment.
Governance enforced with strict security standards maintained across all researcher interactions with Boehringer Ingelheim’s biobank and clinical trial data.
Enablement of responsible reuse of clinical data to turn insights into discoveries that improve healthcare.
59
Minutes vs. months
to complete target identification
500K+
UK Biobank
participant data analysed
125K+
Genomics England
participant data analysed
270+
Analytical processes
Build into an end-to-end protocol spanning 21 large-scale Nextflow pipelines

Lifebit turns Genomic Data into Personalised Cancer Treatment
“If any research organisation is looking to make the most of their data management and have the impact within their community, connecting with the Lifebit team is a must.”
Grant Roesler
DREAM Platform Administrator
CHALLENGES
Strict trans-national privacy and governance requirements
High cost and risk of transferring datasets
Ensuring adherence to data access agreements
high cost and risk of transferring datasets
Harmonizing distributed molecular and clinico-phenotypic datasets from 9+ CROs
Costly cloud analyses limiting researchers’ ability to run all required studies
OUTCOMES
A flexible data management infrastructure that grows as the cohort and data types grow
From Excel to the Command Line – flexible analytical tooling that caters to all user skill levels
Standardized data assets promote findability and usability of diverse molecular and clinico-phenotypic data
Support every step of the way – guided training session to ensure clincians and researchers alike are able to deliver results for their paitents
30–90% lower cloud costs through large-scale analysis optimisation

100%
OMOP mapping
coverage of molecular and clinico-phenotypic data
3
Clinical sites
onboarded to the DREAM platform
100%
adherence
to US and Canadian data governance requirements
30–90%
Lower cloud costs
through large-scale analysis optimisation
Federation
Securely collaborate on distributed data
with the pioneers of data federation

CHALLENGE:
Clinical & multi-omic data is siloed and inaccessible
97% of life-saving health data is inaccessible and unusable due to sensitivity and fragmentation. Difficultly in accessing, connecting and analyzing large cohorts is limiting scientific discovery and health insights.

Lifebit’s federated data solutions enable researchers to securely access and use distributed clinical and multi-omic data at scale, across networks, by bringing the analysis and computation to where the data resides. Uniquely keeping data in situ enables scalable data research while keeping the data secure and protecting patient privacy.
SOLUTION:
Securely connect, access & analyze distributed data in situ
Federation
Separate research databases
successfully federated in what is a UK first
Lifebit worked with University of Cambridge, NIHR Cambridge BRC & Genomics England to successfully federate analysis of genomic healthcare data for the first time in the UK.
“This technology has the potential to remove the geographical, logistical, and financial barriers associated with moving exceptionally large datasets. Applied at scale, this means huge potential for new discoveries, particularly for research into rare diseases and for reducing health inequalities.”
Professor Serena Nik-Zainal, University of Cambridge
Resources
Insights from the world of health data.
Case studies, news and research updates.
Ready to
transform your research?
See how Lifebit can accelerate your research while keeping your data secure and compliant











